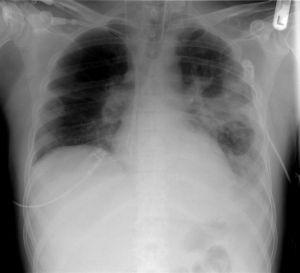
Pneumonia (main)
This page is for adult patients. For pediatric patients, see: pneumonia (peds)
Background
- Definition: infection of lung parenchyma
- Most common infectious cause of death in the United States; >1.5 million adults hospitalized annually for CAP[1]
- Up to 50% of cases have no pathogen identified even with extensive workup[1]
- Empirically classified based upon location/risk factors
Pneumonia Empiric Categories
The term "health care-associated pneumonia" (HCAP) is no longer used.[2] It previously referred to pneumonia acquired in any healthcare facility (e.g., nursing home, hemodialysis center, recent hospitalization) and was used to identify patients at risk for infection with multidrug-resistant pathogens. However, this inappropriately led to increased inappropriately broad antibiotic use and was thus retired. Patients previously classified as HCAP should in general be treated as CAP with exceptions as below under resistant pathogens.
- Community-acquired pneumonia (CAP): Acquired outside of the hospital
- Nosocomial pneumonia: Acquired in a hospital setting
- Hospital-acquired pneumonia (HAP): Acquired ≥48 hours after hospital admission
- Ventilator-associated pneumonia (VAP): Acquired ≥48 hours after endotracheal intubation
Resistant Pathogen Risk Factors
IDSA recommends covering empirically for resistant pathogens (e.g., MRSA, pseudomonas) in adults with CAP only if there is a treatment regimen based on "locally validated" risk factors. In that case, may give empiric coverage while awaiting culture results.[2]
- Commonly accepted risk factors historically include:
- Prior respiratory isolation of MRSA or P. aeruginosa
- Recent hospitalization AND receipt of parenteral antibiotics (in the last 90 days)
- Nursing home/long-term care residents
- Dialysis
- Receiving chronic wound care
- Receiving chemotherapy
- Immunosuppression (including steroids)
- Alcoholism
- Structural lung disease
- Malnutrition
Commonly Encountered Pathogens by Risk Factor
| Risk Factor | Associated Organism |
| Alcoholism | |
| *Aspiration | |
| COPD and/or Smoking | |
| Nursing Home | |
| Exposure to bird droppings | |
| Exposure to birds | |
| Exposure to rabbits | |
| Exposure to farm animals |
|
| Exposure to southwestern US |
|
| Early HIV | |
| Late HIV (as above, plus:) | |
| Structural Lung Disease (CF, bronchiectasis) | |
| Injection drug use | |
| Influenza |
|
| Ventilator Associated Pneumonia |
Causes of Pneumonia
Bacteria
Viral
- Common
- Influenza
- Respiratory syncytial virus
- Parainfluenza
- Rarer
- Adenovirus
- Metapneumovirus
- Severe acute respiratory syndrome (SARS)
- Middle east respiratory syndrome coronavirus (MERS)
- 2019-nCoV (COVID-19)
- Cause other diseases, but sometimes cause pneumonia
Fungal
- Histoplasmosis
- Coccidioidomycosis
- Blastomycosis
- Pneumocystis jirovecii pneumonia (PCP)
- Sporotrichosis
- Cryptococcosis
- Aspergillosis
- Candidiasis
Parasitic
Clinical Features
- Fever, chills, pleuritic chest pain, productive cough
- Fever is seen in 80%
- Tachypnea
- Most sensitive sign in elderly
- Abdominal pain, nausea and vomiting, diarrhea may be seen with Legionella infection
- Myalgia, fatigue
ED Pearls
- Elderly/immunocompromised patients may present atypically: altered mental status, falls, functional decline, or hypothermia rather than classic fever and cough[3]
- Tachypnea (RR >24) is the most sensitive vital sign finding in elderly patients and should prompt evaluation even in the absence of typical symptoms
- Sepsis screening should be performed in all patients with suspected pneumonia; pneumonia is the most common cause of sepsis
- Patients with lower lobe pneumonia may present with predominantly upper abdominal pain, mimicking an acute abdomen
Differential Diagnosis
Acute dyspnea
Emergent
- Pulmonary
- Airway obstruction
- Anaphylaxis
- Angioedema
- Aspiration
- Acute respiratory distress syndrome (ARDS)
- Asthma
- Cor pulmonale
- Epiglottitis
- Inhalation exposure
- Noncardiogenic pulmonary edema
- Pneumonia
- Pneumocystis Pneumonia (PCP)
- Pulmonary embolism
- Pulmonary hypertension
- Tension pneumothorax
- Idiopathic pulmonary fibrosis acute exacerbation
- Cystic fibrosis exacerbation
- Cardiac
- Other Associated with Normal/↑ Respiratory Effort
- Other Associated with ↓ Respiratory Effort
Non-Emergent
- ALS
- Ascites
- Uncorrected ASD
- Congenital heart disease
- COPD exacerbation
- Deconditioning
- Fever
- Hyperventilation
- Interstitial lung disease
- Neoplasm
- Obesity
- Obstructive sleep apnea
- Panic attack
- Pleural effusion
- Polymyositis
- Porphyria
- Pregnancy
- Rib fracture
- Spontaneous pneumothorax
- Thyroid Disease
- URI
- Vocal cord dysfunction
Evaluation
Workup
- CXR
- May have negative CXR early in disease or in cases of dehydration; infiltrate may "blossom" after providing rehydration and repeat imaging[3]
- Absence of CXR findings does not preclude diagnosis; high clinical suspicion with adventitious breath sounds can be consistent with pneumonia despite negative imaging
- Immunocompromised patients may not manifest radiographic evidence of pneumonia despite suggestive clinical findings
- Clinical and radiographic findings do not necessarily correspond: the patient may be improving clinically despite having a worsening appearance on the CXR
- Ultrasound
- Can be considered as an alternative to CXR
- Sensitivity 82% and specificity 94%[4]
- Key findings: hepatization of lung tissue, dynamic air bronchograms, B-lines, and subpleural consolidation
- Particularly useful for identifying parapneumonic pleural effusion
- CBC
- Chemistry (including BUN, glucose for PSI scoring)
- Lactate if concern for sepsis
- IDSA does not support using initial serum procalcitonin levels to determine whether empiric antibiotics should be initiated[2]
- Clinical judgement plus radiographic evidence alone should guide therapy (strong recommendation, moderate quality of evidence)
- Procalcitonin sensitivity for detecting bacterial infection ranges from 38% to 91%; not accurate enough to justify withholding antibiotics[5]
If patient will be admitted:
- Blood Cultures are ONLY indicated for CAP patients with:[2]
- ICU admission (required)
- Empiric treatment for MRSA or P. aeruginosa (required)
- Prior MRSA or P. aeruginosa respiratory infection
- Recent hospitalization AND parenteral antibiotics in the last 90 days
- Multi-lobar infiltrates
- Pleural effusion
- Cavitary lesions
- Leukopenia
- The yield of blood cultures is low (2% to 9%) in adults without severe CAP, and empiric therapy is rarely changed based on these results[5]
- Sputum Gram stain and culture: recommended for the same indications as blood cultures above[2]
- Lower respiratory tract samples from intubated patients should be sent for Gram stain and culture shortly after intubation (higher yield than sputum)
- MRSA nasal PCR (if available): High negative predictive value (~95%) for ruling out MRSA pneumonia; useful for de-escalation of empiric MRSA coverage[6]
- Legionella and Pneumococcal urinary antigen testing: Not routinely recommended; consider for:[2]
- Severe CAP (ICU patients)
- Legionella antigen in alcoholics, outbreaks, or recent (within 2 weeks) travel history
- Influenza testing: Recommended when influenza is circulating in the community; molecular testing (PCR) preferred over rapid antigen[2]
Chest X-Ray Mimics
- Malignancy
- Tuberculosis
- Pulmonary embolism - Hampton's hump
- Pleural effusion
- Atelectasis
- ARDS
- Diffuse alveolar hemorrhage
- Multiple "cannonball" infiltrates
- Metastatic disease
- Septic emboli
- Right sided endocarditis
IDSA Severe Pneumonia Criteria
Severe pneumonia can be diagnosed with either one major criterion or three or more minor criteria.[7] These criteria have a pooled sensitivity of 84% and specificity of 78% for predicting ICU admission.[2]
Minor criteria
- Respiratory rate > 30 breaths/min
- PaO2/FiO2 ratio < 250
- Multi-lobar infiltrates
- Confusion/disorientation
- Uremia (blood urea nitrogen level > 20 mg/dl)
- Leukopenia (WBC < 4,000 cells/µL)
- Thrombocytopenia (platelet count < 100,000 cells/µL)
- Hypothermia (<36°C)
- Hypotension requiring aggressive fluid resuscitation
Major criteria
- Septic shock with need for vasopressors
- Respiratory failure requiring mechanical ventilation
Management
Outpatient
Coverage targeted at S. pneumoniae, H. influenzae, M. pneumoniae, C. pneumoniae, and Legionella
Healthy[2]
No comorbidities (chronic heart, lung, liver, or renal disease; diabetes mellitus; alcoholism; malignancy; or asplenia) and no risk factors for MRSA or Pseudomonas aeruginosa (include prior respiratory isolation of MRSA or P. aeruginosa or recent hospitalization AND receipt of parenteral antibiotics (in the last 90 d))
- Amoxicillin 1 g three times daily (strong recommendation, moderate quality of evidence), OR
- Doxycycline 100 mg twice daily (conditional recommendation, low quality of evidence), OR
- Macrolide in areas with pneumococcal resistance to macrolides <25% (conditional recommendation, moderate quality of evidence).
- Azithromycin 500 mg on first day then 250 mg daily OR
- Clarithromycin 500 mg BID or clarithromycin ER 1,000 mg daily
- Duration of therapy 5 days minimum
Unhealthy[2]
If patient has comorbidities or risk factors for MRSA or Pseudomonas aeruginosa
- Combination therapy:
- Amoxicillin/Clavulanate
- 500 mg/125 mg TID OR amox/clav 875 mg/125 mg BID OR 2,000 mg/125 mg BID. Duration is for a minimum of 5 days and varies based on disease severity and response to therapy; patients should be afebrile for ≥48 hours and clinically stable before therapy is discontinued[8]
- OR cephalosporin
- Cefpodoxime 200 mg BID OR cefuroxime 500 mg BID
- AND macrolide
- Azithromycin 500 mg on first day then 250 mg daily
- OR clarithromycin 500 mg BID OR clarithromycin ER 1,000 mg daily (strong recommendation, moderate quality of evidence for combination therapy)
- OR doxycycline 100 mg BID (conditional recommendation, low quality of evidence for combination therapy)
- Amoxicillin/Clavulanate
- Monotherapy: respiratory fluoroquinolone (strong recommendation, moderate quality of evidence):
- Levofloxacin 750 mg daily OR
- Moxifloxacin 400 mg daily OR
- Gemifloxacin 320 mg daily
Inpatient
- Monotherapy or combination therapy is acceptable
- Combination therapy includes a cephalosporin and macrolide targeting atypicals and Strep Pneumonia[9]
- Adjunctive corticosteroids in severe CAP: The SCCM 2024 Focused Update strongly recommends corticosteroids for hospitalized adults with severe bacterial CAP (strong recommendation, moderate certainty)[10]
- CAPE COD trial (NEJM 2023): Hydrocortisone 200 mg IV daily (50 mg q6h) in severe CAP requiring ICU/intermediate care → ↓ 28-day mortality (6.2% vs 11.9%, NNT ~18), ↓ intubation, ↓ vasopressor use[11]
- Duration: 200 mg/day for 4–7 days based on clinical improvement, then tapered (total 8–14 days)
- Excluded patients already in septic shock
- No recommendation for or against steroids in less severe CAP[10]
- Avoid in influenza pneumonia (without bacterial superinfection)[2]
- Duration: Minimum 5 days; continue until clinically stable (temp ≤37.8°C, HR ≤100, RR ≤24, SBP ≥90, SpO2 ≥90% on RA, tolerating PO, baseline mental status) for ≥48 hours[2]
- De-escalation: If empiric MRSA or Pseudomonas coverage was started, de-escalate to standard CAP therapy within 48 hours if cultures/MRSA nasal PCR are negative and patient is improving[2]
Community Acquired (Non-ICU)
Coverage against community acquired organisms plus M. catarrhalis, Klebsiella, S. aureus[2]
- β-lactam (e.g. ceftriaxone 1–2g daily OR ampicillin-sulbactam 1.5–3g q6h OR cefotaxime 1–2g q8h OR ceftaroline 600mg q12h) PLUS
- Macrolide (e.g. azithromycin 500 mg daily or clarithromycin 500 mg BID) OR
- Doxycycline 100mg IV/PO BID (if contraindications to both macrolides and fluoroquinolones) OR
- Levofloxacin 750mg IV/PO once daily OR
- Moxifloxacin 400mg IV/PO once daily
ICU, Low Risk of MRSA/Pseudomonas
- Ceftriaxone 1-2g IV + Azithromycin 500mg IV OR
- Ceftriaxone 1-2g IV + (moxifloxacin 400mg IV or levofloxacin 750mg IV)
- Penicillin allergy:
- (Moxifloxacin or levofloxacin) + (aztreonam 1-2g IV or clindamycin 600mg IV)
ICU, Risk of Pseudomonas (without MRSA risk)
2019 guidelines recommend single antipseudomonal β-lactam (changed from double gram-negative coverage in 2007 guidelines)[2]
- Antipseudomonal β-lactam: Piperacillin-Tazobactam 4.5g q6h OR Cefepime 2g q8h OR meropenem 1g q8h OR Imipenem 500mg q6h
- PLUS azithromycin 500mg IV daily OR respiratory fluoroquinolone (levofloxacin 750mg IV daily or moxifloxacin 400mg IV daily)
- If Pseudomonas is not isolated and patient is improving, de-escalate to standard CAP regimen[2]
ICU, Risk of MRSA
Add MRSA coverage to appropriate regimen above[2]
- Vancomycin 15–20 mg/kg IV q8-12h (target AUC/MIC 400-600) OR Linezolid 600 mg IV q12h
- MRSA nasal PCR has a high negative predictive value (~95%); if negative, MRSA coverage can be safely discontinued[12]
Hospital Acquired Pneumonia (HAP)
Pneumonia developing ≥48 hours after hospital admission in non-intubated patients[13]
- High risk of MRSA or high mortality risk (ventilatory support for HAP or septic shock)
- Antipseudomonal β-lactam from two different classes with activity against Pseudomonas:
- Piperacillin-Tazobactam 4.5g q6h OR Cefepime 2g q8h OR ceftazidime 2g q8h OR meropenem 1g q8h OR Imipenem 500mg q6h
- PLUS antipseudomonal non-β-lactam: Levofloxacin 750mg IV q24h OR ciprofloxacin 400mg q8h OR aminoglycoside (e.g. tobramycin, gentamicin, amikacin)
- PLUS Vancomycin 15-20 mg/kg IV q8-12h OR Linezolid 600mg IV q12h
- Low risk of MRSA and low mortality risk
- Single antipseudomonal β-lactam (from list above) may be sufficient[13]
- Of note, the combination of vancomycin + piperacillin-tazobactam carries higher risk of AKI compared to cefepime + vancomycin[14]
- Consider tobramycin or other aminoglycoside in place of fluoroquinolones given FDA 2016 warnings
- Duration: 7 days recommended for HAP/VAP[13]
Ventilator Associated Pneumonia (VAP)
Pneumonia developing ≥48 hours after endotracheal intubation[13]
- High risk of MRSA or IV antibiotics in the last 90 days or unit MRSA prevalence >10-20% or unknown
- Include an antibiotic from each of these 3 categories:
- 1. MRSA coverage: Vancomycin 15-20 mg/kg IV q8-12h OR Linezolid 600 mg IV q12h PLUS
- 2. Antipseudomonal β-Lactam: Piperacillin-Tazobactam 4.5g q6h OR Cefepime 2g q8h OR meropenem 1g q8h OR Imipenem 500mg q6h OR Aztreonam 2g q8h PLUS
- 3. Antipseudomonal non-β-Lactam: Ciprofloxacin 400mg IV q8h OR Levofloxacin 750mg IV q24h OR aminoglycoside
- Low risk of MRSA and Pseudomonas (no risk factors for antimicrobial resistance, unit MRSA <10-20%)
- Single antipseudomonal β-lactam monotherapy (from list above) is acceptable[13]
- Duration: 7 days recommended[13]
Adjunctive Corticosteroids
Updated guidance based on CAPE COD trial (2023) and SCCM 2024 Focused Update:
- The SCCM 2024 Focused Update strongly recommends corticosteroids for adult patients hospitalized with severe bacterial CAP (strong recommendation, moderate certainty of evidence)[10]
- No recommendation for or against steroids in less severe CAP
- The CAPE COD trial (NEJM 2023): IV hydrocortisone 200 mg/day in severe CAP requiring ICU/intermediate care admission showed:[11]
- ↓ 28-day mortality (6.2% vs 11.9%; absolute difference 5.6%; NNT ~18)
- ↓ Need for intubation
- ↓ Need for vasopressors
- Duration: hydrocortisone 200 mg/day for 4 or 7 days based on clinical improvement, then tapered (total 8 or 14 days)
- Excluded patients already in septic shock
- REMAP-CAP (2025): Fixed-dose hydrocortisone 200 mg/day x 7 days in severe CAP triggered a futility stopping rule for 90-day mortality; however, this trial enrolled a more severely ill population with higher rates of septic shock than CAPE COD[15]
- Practical approach for the ED:
- If patient has severe CAP (meets IDSA/ATS severity criteria, hypoxic, or being admitted to the ICU), consider hydrocortisone 200 mg IV daily (50 mg q6h) or methylprednisolone 40 mg IV daily
- No clear benefit in non-severe CAP
- Avoid in active influenza pneumonia (without bacterial superinfection) per 2019 ATS/IDSA[2]
Aspiration Pneumonia
See also: Aspiration pneumonia and pneumonitis
- 2019 ATS/IDSA guidelines recommend against routinely adding anaerobic coverage beyond standard CAP empiric antibiotics in suspected aspiration pneumonia (conditional recommendation, very low quality of evidence)[2]
- Exception: Add anaerobic coverage (e.g. ampicillin-sulbactam, clindamycin, or carbapenem) if lung abscess or empyema is suspected
- Rationale: More recent studies show anaerobes are uncommon causes of aspiration pneumonia; unnecessary anaerobic coverage increases risk of Clostridioides difficile infection without mortality benefit[16]
- Aspiration pneumonitis (chemical/inflammatory reaction without infection): supportive care only; antibiotics are not indicated unless infection develops
Influenza-Associated Pneumonia
- Anti-influenza treatment (e.g. oseltamivir) should be prescribed to all adults with CAP who test positive for influenza, regardless of duration of illness or hospitalization status[2]
- Standard antibacterial treatment should also be initially prescribed alongside antivirals in patients with clinical and radiographic evidence of CAP who test positive for influenza[2]
- Avoid routine corticosteroids in influenza pneumonia[2]
Treatment Duration and De-escalation
- Minimum 5 days of antibiotics[2]
- Continue until the patient achieves clinical stability, defined as:
- Temperature ≤37.8°C
- Heart rate ≤100 bpm
- Respiratory rate ≤24
- Systolic BP ≥90 mmHg
- SpO2 ≥90% on room air
- Able to take oral intake
- Normal mental status (back to baseline)
- De-escalation: If empiric MRSA or Pseudomonas coverage was started, de-escalate to standard CAP therapy within 48 hours if cultures and MRSA nasal PCR are negative and the patient is improving[2]
Treatment Failure
Consider the following if patient is not improving at 48-72 hours:
- Incorrect diagnosis (see CXR Mimics above)
- Resistant or unusual organism (obtain cultures if not already done)
- Complications: empyema, lung abscess, parapneumonic effusion
- Inadequate antibiotic dosing or penetration
- Superinfection (e.g. C. difficile)
- Immunocompromised state not initially recognized
- Noninfectious mimic (e.g. pulmonary embolism, malignancy)
- Consider CT chest for complications not visible on CXR
Disposition
- Use clinical judgment plus PORT Score (PSI) to guide disposition per IDSA 2019 guidelines[2]
- Discharge (PSI class I-II):
- Able to tolerate oral medications
- No hypoxia, hemodynamic instability, or significant comorbidities
- Reliable follow-up in 48-72 hours
- Admit to floor (PSI class III-IV):
- Hypoxia requiring supplemental O2
- Inability to tolerate oral medications
- Significant comorbidity (immunocompromised, decompensated CHF, etc.)
- Admit to ICU (PSI class V, or meeting major/minor criteria):
- Requiring vasopressors or mechanical ventilation (major criteria)
- 3 or more minor criteria: RR >30, PaO2/FiO2 <250, multilobar infiltrates, confusion, BUN >20, WBC <4, platelets <100, hypothermia, hypotension requiring aggressive fluids
Prognosis
- Half of patients are still symptomatic at 30 days, with a significant minority experiencing chest pain, malaise or mild dyspnea even 2 to 3 months after treatment
- In adults with CAP whose symptoms have resolved within 5-7 days, it is not recommended to routinely obtain follow-up chest imaging[2]
- Consider follow-up imaging in 7-12 weeks for patients at higher risk for malignancy (e.g. smokers >50 years) to ensure resolution and exclude underlying malignancy
See Also
- Pneumonia (Pathogens)
- Pneumonia (Peds)
- Ventilator associated pneumonia
- Aspiration pneumonia and pneumonitis
- Antibiotics by diagnosis
- CURB-65
- Sepsis (Main)
Calculators
Template:PSI Calculator Template:PORT Score Calculator
CURB-65 Score
| Criteria | No (0) | Yes (+1) |
|---|---|---|
| Confusion (new disorientation in person, place, or time) | 1 | |
| Uremia — BUN >19 mg/dL (>7 mmol/L) | 1 | |
| Respiratory rate ≥30 breaths/min | 1 | |
| Blood pressure — SBP <90 mmHg or DBP ≤60 mmHg | 1 | |
| Age ≥65 years | 1 | |
| CURB-65 Score | / 5 | |
| 0–1 | Low Risk — 1.5% 30-day mortality. Consider outpatient treatment with oral antibiotics. |
|---|---|
| 2 | Moderate Risk — 9.2% 30-day mortality. Consider short inpatient stay or closely monitored observation. |
| 3–5 | High Risk — 22% 30-day mortality. Inpatient admission recommended. ICU if score 4–5. |
| References |
|---|
|
PSI/PORT Score
| Demographics | Value |
|---|---|
| Age (years) | |
| Female sex (−10 points) | 1 No Yes |
| Comorbidities | Points |
| Nursing home resident (+10) | 1 No Yes |
| Neoplastic disease (+30) | 1 No Yes |
| Liver disease (+20) | 1 No Yes |
| CHF (+10) | 1 No Yes |
| Cerebrovascular disease (+10) | 1 No Yes |
| Renal disease (+10) | 1 No Yes |
| Physical Exam | Points |
| Altered mental status (+20) | 1 No Yes |
| Respiratory rate ≥30 (+20) | 1 No Yes |
| Systolic BP <90 (+20) | 1 No Yes |
| Temperature <35 or ≥40°C (+15) | 1 No Yes |
| Pulse ≥125 (+10) | 1 No Yes |
| Labs/Imaging | Points |
| pH <7.35 (+30) | 1 No Yes |
| BUN ≥30 (+20) | 1 No Yes |
| Sodium <130 (+20) | 1 No Yes |
| Glucose ≥250 (+10) | 1 No Yes |
| Hematocrit <30% (+10) | 1 No Yes |
| PaO₂ <60 (+10) | 1 No Yes |
| Pleural effusion (+10) | 1 No Yes |
| PSI Score |
| ≤50 (Class I) | 0.1% mortality — Outpatient treatment. |
|---|---|
| 51–70 (Class II) | 0.6% mortality — Outpatient treatment. |
| 71–90 (Class III) | 2.8% mortality — Brief inpatient observation. |
| 91–130 (Class IV) | 8.2% mortality — Inpatient treatment. |
| >130 (Class V) | 29.2% mortality — Inpatient/ICU. |
|
External Links
- MDCalc - CURB-65 Severity Score
- MDCalc - PSI/PORT Score
- ATS/IDSA 2019 CAP Guidelines (Full Text)
- IDSA/ATS 2016 HAP/VAP Guidelines (Full Text)
References
- ↑ 1.0 1.1 Jain S, Self WH, Wunderink RG, et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med. 2015;373(5):415-427.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 2.11 2.12 2.13 2.14 2.15 2.16 2.17 2.18 2.19 2.20 2.21 2.22 2.23 2.24 2.25 Diagnosis and Treatment of Adults with Community-acquired Pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America Am J Respir Crit Care Med. 2019 Oct 1;200(7):e45-e67
- ↑ 3.0 3.1 Feldman C. Pneumonia in the elderly. Clin Chest Med. 1999;20(3):563-573. doi:10.1016/s0272-5231(05)70236-7
- ↑ Staub LJ, Mazzali Biscaro RR, Kaszubowski E, Maurici R. Lung Ultrasound for the Emergency Diagnosis of Pneumonia, Acute Heart Failure, and Exacerbations of Chronic Obstructive Pulmonary Disease/Asthma in Adults: A Systematic Review and Meta-analysis. J Emerg Med. 2019;56(1):53-69. doi:10.1016/j.jemermed.2018.09.009
- ↑ 5.0 5.1 Metlay JP, Waterer GW. Update on Community-Acquired Pneumonia: Updated Recommendations from the ATS and IDSA. Am Fam Physician. 2020;102(2):120-121.
- ↑ Parente DM, Cunha CB, Engemann AM, et al. The Clinical Utility of Methicillin-Resistant Staphylococcus aureus (MRSA) Nasal Screening to Rule Out MRSA Pneumonia: A Diagnostic Meta-analysis With Antimicrobial Stewardship Implications. Clin Infect Dis. 2018;67(1):1-7.
- ↑ Severe pneumonia can be diagnosed with either one major criterion or three or more minor criteria. Neth J Med. 1999 Sep;55(3):110-7 10.1016/s0300-2977(99)00071-6
- ↑ IDSA. Mandell 2007
- ↑ Chokshi R, Restrepo MI, Weeratunge N, Frei CR, Anzueto A, Mortensen EM. Monotherapy versus combination antibiotic therapy for patients with bacteremic Streptococcus pneumoniae community-acquired pneumonia. Eur J Clin Microbiol Infect Dis. Jul 2007;26(7):447-51
- ↑ 10.0 10.1 10.2 Chaudhuri D, Nei AM, Rochwerg B, et al. 2024 Focused Update: Guidelines on Use of Corticosteroids in Sepsis, Acute Respiratory Distress Syndrome, and Community-Acquired Pneumonia. Crit Care Med. 2024;52(5):e219-e233.
- ↑ 11.0 11.1 Dequin PF, Meziani F, Quenot JP, et al. Hydrocortisone in Severe Community-Acquired Pneumonia. N Engl J Med. 2023;388(21):1931-1941.
- ↑ Parente DM, Cunha CB, Engemann AM, et al. The Clinical Utility of Methicillin-Resistant Staphylococcus aureus (MRSA) Nasal Screening to Rule Out MRSA Pneumonia: A Diagnostic Meta-analysis With Antimicrobial Stewardship Implications. Clin Infect Dis. 2018;67(1):1-7.
- ↑ 13.0 13.1 13.2 13.3 13.4 13.5 Kalil AC, Metersky ML, Klompas M et al. Management of Adults With Hospital-acquired and Ventilator-associated Pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016 Sep 1;63(5):e61-e111.
- ↑ Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL. Vancomycin Plus Piperacillin-Tazobactam and Acute Kidney Injury in Adults: A Systematic Review and Meta-Analysis. Crit Care Med. 2018;46(1):12-20.
- ↑ REMAP-CAP Investigators. Effect of hydrocortisone on mortality in patients with severe community-acquired pneumonia. Intensive Care Med. 2025.
- ↑ Tessier L, Bhatt M, Engert JC, et al. Anaerobic Antibiotic Coverage in Aspiration Pneumonia and the Associated Benefits and Harms: A Retrospective Cohort Study. Chest. 2024;166(3):488-497.